-
 Biliary interventions: Current treatment of Benign biliary and postoperative anastomotic strictures
Biliary interventions: Current treatment of Benign biliary and postoperative anastomotic stricturesBiliary interventions: Current treatment of Benign biliary and postoperative anastomotic strictures● DATE: 19th January, 2021● TIME: CST 05:00, CET 12:00, AET 22:00, KST/JST 20:00● MODERATOR: Arthur Kaffes (Australia), Dong Ki Lee (Korea)● LECTURE: Tatsuya Sato (Japan), Deepak Joshi (UK), José G. de la Mora-Levy (Mexico)● LECTURE ABOUT:- Tatsuya Sato (Japan) "Outcomes of management for benign hepatico jejunostomy stricture"- Deepak Joshi (UK) "Endoscopic treatment of Benign biliary Stricture & Post L.T.A.S."- José G. de la Mora-Levy (Mexico) "Less common and potential future use of the Kaffes Stent"● REGISTER LINK: bit.ly/KaffesWebinar_W
20.12.29 -
 Lectures for EUS-guided RFA, EUSRA
Lectures for EUS-guided RFA, EUSRALectures for EUS-guided RFA, EUSRA● DATE: 7th January, 2021● TIME: 1PM CET● INVITED PANEL: Dongwan Seo (Korea), Stephen Pereira (UK), Pradermchai Kongkam (Thailand), Nirav Thosani (USA)● LECTURE ABOUT:- Dongwan Seo (Korea) "EUS-RFA of pancreatic cystic tumor"- Stephen Pereira (UK) "EUS-RFA of benign pancreatic and adrenal tumors"- Pradermchai Kongkam (Thailand) "EUS-RFA of Unresectable pancreatic cancer"- Nirav Thosani (USA) "EUS-RFA of borderline resectable pancreatic cancer"● REGISTER LINK: https://bit.ly/EUS-RFA_W
20.12.15 -
 CONNECT GLOBAL issue 10, 2019 19.10.30
CONNECT GLOBAL issue 10, 2019 19.10.30 -
 United European Gastroenterology Week 2019
United European Gastroenterology Week 2019United European Gastroenterology Week 2019UEGW 2019 was held in Barcelona, Spain from October 21st to 23rd. Taewoong Medical participated as an exhibitor and opened a 40 sqm peninsula booth. The booth was decorated with the newly launched product, Niti-S™ HOT SPAXUS™. One day before the exhibition, Taewoong Medical invited their sales representatives to a Partners’ Day event and shared the Product News including the new launching of HOT SPAXUS™ and EUSTAR™ FNA needle. Thanks to Mr. Hiroshi Honoki and Mr. Doug Polzin’s sales experience sharing, the meeting was very productive and vibrant. On the 21st, Taewoong Medical held the 9th Taewoong Medical Dinner Symposium and enjoyed the evening with intensive lectures by Prof. Anthony Teoh and Prof. Carlos de la Serna.
19.10.30 -
 Digestive Disease Week 2019
Digestive Disease Week 2019Digestive Disease Week 2019The Digestive Disease Week® (DDW) 2019 annual meeting took place in San Diego, the beautiful city with a pacific shore of the US west coast was the host for the 50th anniversary of the DDW. Taewoong Medical participated in the meeting for the 13th consecutive time and held one of the major booths.Since ELRA™ received FDA approvals recently, all RFA product lines became available in the US market. Both EUSRA™, the needle type EUS-RFA probe for pancreatic tumors, and ELRA™, the bipolar endo-biliary RFA probe for hilar and the common bile duct received extensive attention from the attendees. In addition, new abstracts regarding ELRA™ and EUSRA™ results in various indications were also published. We appreciate for your visits and interests. We look forward to further feedbacks regarding our products.See you all next year!
19.05.30 -
 ESGE Days 2019
ESGE Days 2019ESGE Days 2019 ESGE Days 2019, which was the second exhibition attended this year, started last year in Budapest. Building on the innovative ESGE Days programme developed for first congress, in Prague for ESGE Days 2019. The interactive, comprehensive programme will reflect differences in terms of delegate experience levels and will also be relevant to all members of the endoscopy team (nurses, residents, fellows, physicians).The number of doctors registered this year was 2,667, twice as many as last year. The greatest number of physicians came from Czech Republic, Spain, Germany, Italy, France, UK, Netherlands and Korea. ESGE Days predicted that the number of registration will increase every year. Following the UEGW, the ESGE Days is expected to become a society representing the gastrointestinal endoscopy society in Europe.We were able to feel that EUSRA™ was increasingly interested in the EUS therapeutic pancreas sessions almost 50% of lectures are related with Taewoong Medical. In the future, Taewoong Medical will do our best to meet various physician who interested in our product on the international congress. Next year ESGE Days will be held on April 23-25 in Dublin, Ireland.
19.04.24 -
 Asian Pacific Digestive Week 2018
Asian Pacific Digestive Week 2018Asian Pacific Digestive Week 2018 This year’s APDW was held on November 15th, 2018 in Taewoong’s Niti-S™ Stent home country of South Korea at the grand venue of COEX Convention Center in Seoul. It was a meaningful time as Taewoong was able to participate in the conference and connect with many of our clients. Before the conference, 50 clients from 14 difference countries were able to visit our Headquarters to observe the production process of our handmade stents while also being able to resolve all of the questions they had. In addition, Taewoong’s products were able to be known more to our clients through the EUSRA Live case done by Prof. Seo Dong Wan of Asan Hospital and the Hands On Stenting course in the Hilar Obstruction using the Niti-S LCD™ and the Optimos™ Sphiecterotome done by Prof. Yang Min Jae of Aju Hospital and Prof. Jeon Tae Ju of Injae Hospital.
18.11.23 -
 United European Gastroenterology Week 2018
United European Gastroenterology Week 2018United European Gastroenterology Week 2018 ● Date: Oct 22nd to 24th, 2018● Venue: Austria Center Vienna, Vienna, Austria● Booth Exhibition & Taewoong Medical Dinner Symposium The 26th United European Gastroenterology Week (UEGW) was held at Austria Center Vienna from October 22nd to the 24th. As always, Taewoong Medical participated as an exhibitor and enjoyed lively interactions between industries and doctors. The newly released products including the Biliary S Flare stent and the Esophageal Dual stent were exposed to the public for the first time and achieved huge attention from the attendees. In addition, the EUSRA RFA device which was approved by the US FDA right before the congress also drew extensive interest.To measure up the high interest in the advanced EUS-guided procedure, Taewoong Medical prepared a Dinner symposium under the theme of "EUS-guided stenting and radio frequency ablation." For this special event, four prominent experts were invited and they shared their extracted know-hows to the guests.
18.11.05 -
 SNUH’s artificial heart valve wins regulators’ approval
SNUH’s artificial heart valve wins regulators’ approvalSNUH’s artificial heart valve wins regulators’ approval Seoul National University Hospital (SNUH) said Thursday that it has received sales approval from the Ministry of Food and Drug Safety for its pulmonary artery artificial heart valve. The hospital has been researching the device along with TaeWoong Medical since 2014.The research team, led by Professors Kim Ki-bum, Kim Yong-jin and Im Heung-gook at the hospital, began the development of artificial heart valves using pig and bovine adrenal glands with the support from the long-term biosimilar project team funded by the Ministry of Health and Welfare. The team has also developed a stent that does not require patients to undergo an open chest surgery.After completing animal research, it conducted a clinical trial by transplanting the artificial heart valve on 10 patients and followed up on them for six months to prove the device’s effectiveness and safety in 2016.As a result, the patient did not require any immunosuppressant as the valve showed almost no immune rejection, which is the biggest problem of xenotransplantation. Professor Kim Ki-bum “Currently, there are inquiries about the commercialization of the product not only from Asian countries such as Japan, Taiwan, and Hong Kong but Europe,” the hospital said. “The research team will meet with 11 pediatric cardiac centers in six European countries next month and will begin clinical trials early next year.”The heart has four valves that control blood circulation. The most common valve disease is valve stenosis of the aorta. However, TAVI, an aorta artificial heart valve, developed by advanced countries such as the U.S. has already secured most of the market share.The hospital expects to have similar benefits, as the pulmonary artery artificial heart valve developed by SNUH research team is the first commercialized product of its kind in the world. Until now, Korea, the United States and China have been fiercely competing to develop the product.The valve and stent developed by SNUH do not require open chest surgery and has a firm stent that can match the size of the patient’s pulmonary artery. The hospital expects that such benefits can lower the financial burden on patients.”Currently, additional clinical trials are underway at several hospitals in Korea,” Professor Kim Ki-bum said. “If the hospital manages to receive a European CE certification after clinical trials in Europe, it will contribute to the improvement of patient's quality of life and globalization of Korean medical technology.”corea022@docdocdoc.co.kr<© Korea Biomedical Review, All rights reserved.>http://www.koreabiomed.com/news/articleView.html?idxno=4414
18.11.05 -
 CONNECT GLOBAL issue 9, 2018 18.11.05
CONNECT GLOBAL issue 9, 2018 18.11.05 -
 Digestive Disease Week 2018
Digestive Disease Week 2018Digestive Disease Week 2018 ● From: June 2nd to June 5th, 2018● Location: Walter E. Washington Convention Center, Washington DC● Booth Exhibition & Taewoong Medical’s Dinner Party Breaking News that will change the course of the US Market and open a New Era. The Digestive Disease Week® (DDW) 2018 annual meeting took place in Washington, D.C., from June 2nd to the 5th. Taewoong Medical, as we have been doing during the past 12 consecutive years, participated in the largest Gastrointestinal Medical event in USA as an exhibitor with great honor.Lumen-Apposing Metallic Stents (LAMS) such as Taewoong’s own SPAXUS™ and NAGI™ stents, and Endoscopic Radio Frequency Ablation (Endoscopic RFA) devices such as EUSRA™ and ELRA™ received extensive attention from the congress attendees throughout the entire event this year. The fever of this year’s interest and joy created through the DDW event lasted not only during the exhibition hours, but also all night through the evening as Taewoong enjoyed an impressive evening with its partners and customers from all over the world at the Nationals Park, Washington DC’s MLB (Major League Baseball) Stadium. Last but not least, the announcement of Taewoong and Cook’s partnership in the US market gained a lot of attention from the event attendees as it was the start of a new era that will bring great benefits and development to not only both partners but also to the customers and patients in all. It was a great opportunity to see and feel firsthand the changing trends of the new market at such an enormous medical event and a great pleasure to meet and respond directly to our customers from around the world.
18.06.11 -
 Cook Medical, Taewoong Medical Partner to Distribute Stents in the U.S.
Cook Medical, Taewoong Medical Partner to Distribute Stents in the U.S.Cook Medical, Taewoong Medical Partner to Distribute Stents in the U.S. BLOOMINGTON, Ind.–(BUSINESS WIRE)–Jun 4, 2018–To complement its existing line of metal GI stents, Cook Medical has partnered with Taewoong Medical to distribute a selection of stents into the U.S. The new partnership includes the Niti-S™ Through the Scope (TTS) Esophageal Stent, the flagship product of the South Korean company’s Niti-S Self-Expandable line of metal GI stents. Cook Medical will begin distributing the Taewoong portfolio on November 1, 2018.Esophageal stents are often used in the treatment of esophageal cancer. According to the American Cancer Institute for Cancer Research, there are approximately 17,000 new cases reported annually, the vast majority occurring in men. This year alone, esophageal cancer will claim almost 16,000 lives. “We’re excited to continue our goal to support physicians in their treatment of diseases of the GI tract by adding Taewoong’s stent technologies to our existing Evolution® and Zilver® stent offerings,” said Barry Slowey, president of Cook Winston-Salem and vice president of Cook Medical’s Endoscopy speciality. “In addition to their product offering, we’re honored to collaborate with Taewoong because we share a commitment to provide solutions that deliver patient care through innovation.”Unique to the U.S. market, the Niti-S TTS Esophageal Stent is preloaded in a 10.5 Fr delivery system. This allows deployment through a scope with a conical tip designed to navigate tight strictures and tortuous anatomies.In addition to the Niti-S TTS Esophageal Stent, Cook will also be the U.S. distributor for the Niti-S S Esophageal Stent—a standard esophageal stent—as well as the Niti-S Biliary Stent.“We are very pleased and proud to enter into this partnership with Cook Medical, a leading global medical device company,” said Kyongmin Shin, CEO of Taewoong Medical. “We believe Cook Medical’s philosophy and focus towards continuous innovation in product development is akin to Taewoong Medical’s own philosophy. And I am confident that our partnership will bring the optimal stent solutions to U.S. doctors and patients.”About Cook MedicalSince 1963 Cook Medical has worked closely with physicians to develop technologies that eliminate the need for open surgery. Today we are combining medical devices, biologic materials and cellular therapies to help the world’s healthcare systems deliver better outcomes more efficiently. We have always remained family owned so that we have the freedom to focus on what we care about: patients, our employees and our communities. Find out more at www.cookmedical.com, and for the latest news, follow us on Twitter, Facebook and LinkedIn.About Taewoong MedicalTaewoong Medical Co., Ltd, founded in 1991, is a privately-held company based in Seoul, South Korea. It manufactures a range of medical equipment of its own design: gastrointestinal devices, interventional urology stents and GI accessories. Taewoong is committed to maintaining high levels of innovation, knowledge and technical expertise to deliver solutions to their customers.
18.06.06 -
 United European Gastroenterology Week 2017
United European Gastroenterology Week 2017United European Gastroenterology Week 2017 ● Oct.30 ~ Nov.02, 2017● Congress Venue: Fira Barcelona Gran Via, Barcelona, Spain● Booth Exhibition & Taewoong Medical’s Dinner Symposium The 25th UEGW has ended successfully. This year it was held in Gaudi’s hometown of Barcelona, Spain. Taewoong Medical participated in the exhibition with mainly Gastrointestinal Metal stent and Endoscopic RFA products as well. In particular, this year, the same stent companies participated in 12 venues, and it was able to see how stent competition is getting stronger every each year.Even in the midst of intense competition, Taewoong Medical always strives to show that quality products are always improving. As time goes on, we could feel the growing interest in RFA products. Currently, RFA products are undergoing study in many countries around the world. We are looking forward to seeing you in more countries soon. Many people attended to Taewoong Medical dinner symposium, which is celebrating its 7th anniversary this year. This year, a lecture on “ERCP guided radiofrequency ablation” and “Transmural fenestrations: how to keep the window open?” was held. In addition to the lecture, we prepared special time for who attended our dinner symposium. The Dinner symposium was completed with the participation of the enthusiastic flamenco performances of Spain. Next UEGW will be held at Vienna, Austria from Oct.20th to Oct.24th 2018. We are looking forward to seeing you again next year.
17.11.13 -
 CONNECT GLOBAL issue 8, 2017 17.11.07
CONNECT GLOBAL issue 8, 2017 17.11.07 -
 Digestive Disease Week 2017
Digestive Disease Week 2017Digestive Disease Week 2017 ● May 7~9, 2017● Congress Venue: Mccormick Place, Chicago, U.S.A● Booth Exhibition & Taewoong Medical’s Dinner Party We took part in DDW 2017 which was taken place in Mccormick Place, Chicago, U.S.A. The exhibition booth consisted of not only Stent, Endoscopic RFA, SPAXUS area but multi-entrance, meeting room, sofas and table with phone recharger for visitor’s convenience. Modern and simple design, emphasizing SPAXUS and RFA, caught lots of attention. Second day of DDW, about 180 Doctors and distributors from many countries enjoyed dinner with tremendous view at Taewoong Medical’s Niti-S Cruise Dinner, just like 3 years ago. 4 the Beat, invited band, played music during dinner followed by dance party with the DJ. Next DDW will be held at Washington Convention Center from June 2nd to June 5th 2018. We always welcome you to join our program.
17.05.23 -
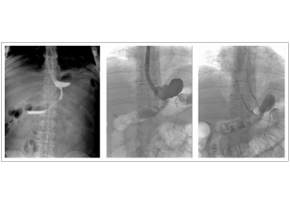 Integrated Approaches for the Management of Staple Line Leaks following Sleeve Gastrectomy
Integrated Approaches for the Management of Staple Line Leaks following Sleeve GastrectomyJ Obes. 2017; 2017: 4703236. Integrated Approaches for the Management of Staple Line Leaks following Sleeve Gastrectomy Endoscopic stenting after staple line leaks has been supported by many authors in recent years, even if this is not a widely accepted treatment. Stent migration is the main complication after the procedure and it has been reported in 20%–59% of cases among different series. In this experience, stents 20 cm long and 24 mm in diameter BETA™ Stents were chosen. In this way, in each case, a good adherence of the stent to the gastric wall. Male, 34 years, BMI 43 Kg/m2. Evidence of leak in radiological upper gastrointestinal series; confirmation of the leak with fluorescence endoscopy; endoscopic gastrooesophageal stenting.
17.04.10
+82 31-904-6196contact@stent.net
HOME
NEWS
News
